Seraccess® SENSE
Unrivalled Artificial Pancreas for Unmanageable Diabetes
Seraccess® was forged from Securecell’s expertise in automating and digitalizing the biotechnology space. Our immense knowledge of connecting expansive bioprocessing systems, microfluidics and documenting processes down to the molecular level led us to solutions that could greatly impact the Artificial Pancreas Device System (APDS) market for a significantly underserved diabetes community: unstable type 1 or 2 diabetes patients.1
Challenges and Solution
How We Change the Game
Current APDS* cannot alleviate the hardship known as unmanageable diabetes.
This disease causes sudden, frequent unpronounced and dramatic swings in blood glucose levels that trigger life-threatening episodes (hypoglycemia or hyperglycemia) due to insufficient precision and time delay of testing and infusing within the subcutaneous tissue. Substantially impaired quality of life and unnecessary excessive economic costs (generated by several emergency interventions per year) keep patients in a constant state of flux from a storm that never quells and uncertainty that never dissipates.
*Artificial Pancreas Device System
Seraccess® SENSE greatly lightens that burden by offering a more effective solution that operates nearly in real time and in direct communication with the patient's bloodstream.
This nearly delay-free, highly accurate and precise blood glucose measurement allows Seraccess® SENSE to administer the exact amount of insulin needed directly into the vein with immediate effect, redirecting unruly blood glucose levels into the target range.**
**ADA guidelines American Diabetes Association
How the Technology Differs
Subcutaneous Testing / Infusion

Seraccess® SENSE

The Problem Seraccess® Solves
Sudden, unpredictable and uncontrollable swings in blood glucose that significantly disrupt almost every aspect of life for those with unmanageable diabetes.
Immediate Complications
- Life Threatening Hypo / Hyperglycemic Attack
- Loss of Consciousness
- Death
Mid / Long-term Complications
- Lower Limb Amputations (Neuropathy)
- Heart Attacks
- Strokes (Cardiovascular Disease)
- Blindness (Retinopathy)
- Kidney Failure
- Significantly Increased Mortality Rate
The only alternative treatments for some of these patients are pancreas or islet cell transplantation (requiring life-long immuno suppressive therapy with severe side effects).
Additionally, these patients suffer from aconstantly disrupted quality of life and greatly impaired personal and social relationships.
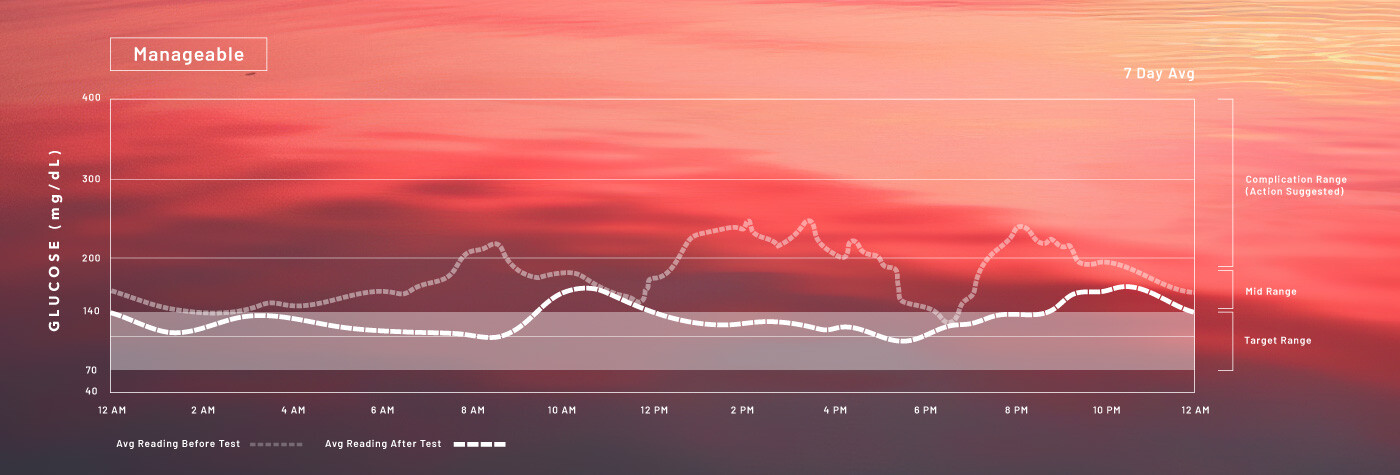
The Daily Torrent
Glucose Sensor Overlay of a young woman with critically unmanageble diabetes. Average glucose swings vastly exceed the Target Range.

Seraccess® SENSE Calms the Storm
Proof of Concept: After a long term control with an intravenous insulin infusion system testing, mean blood glucose concentrations decreased from 22.1 mmol/l to 8.2 mmol/l. With this treatment she could enjoy a “near normal lifestyle”.3
Our Solution
In a first phase, Seraccess® SENSE works with standard catheters, in a later stage with our patented SeraPort catheter developed inhouse.

Benefits
Glucose is measured directly in the blood without delay and insulin is infused directly into the bloodstream resulting in immediate and effective blood glucose control.
This unrivalled procedure mimics the natural pancreatic performance through automated insulin delivery algorithms (AID).
Seraccess® SENSE Key Points:
- Closed-Loop Artificial Pancreas Device System (APDS)
- Fully Automated
- No Patient Input Needed
- Highly Accurate And Precise
- No Blood Loss (Small Blood Sample Aspirated And Flushed Back Into Patient)
- Works With Standard Catheter
- Patent Application Ongoing
SERACCESS® SENSE IS THE ONLY TREATMENT FOR PATIENTS WHERE GOLD STANDARD TREATMENTS FAIL TO MANAGE BLOOD GLUCOSE WITHIN THE TARGET RANGE.
Seraccess® SENSE is a fastacting closed-loop controlsystem that will revolutionizetherapy in the unmanageablediabetes patient group.With your help, we can restorehealth and quality of life tothose with type 1 or 2 diabeteswhile significantly reducing theiroverall health care costs.
Seraccess® SENSE has the capacity to bring so much more than blood glucose back into the Target Range.
Disclaimer
Seraccess® is a mid-stage venture project of Securecell that offers investment opportunities to private and institutional investors.
All information and opinions contained herein have been prepared by management and its advisors and represent their assessment as of January 2022. No representation or warranty, expressed or implied, is given as to the accuracy or completeness of the contents, opinions, or projections expressed herein, and no responsibility or liability is accepted. All information videos and contents do not constitute an offer to purchase securities. The website content does not constitute an invitation to buy shares under US or international law.
References:
1. Hirsch IB et al. A new look at brittle diabetes. Journal of Diabetes and Its Complications 2021;35;1f
2. Leyerla R et al: Recurrent DKA results in high societal costs – a retrospective study identifying social predictors of recurrence for potential future intervention. Clin Diabetes Endocrinol 2021; 7; 13f
3. Bayliss J: Brittle diabetes: long-term control with a portable, continuous, intravenous insulin infusion system. British Medical Journal 1981; 283; 1207f